28 Dec Intranasal COVID-19 Vaccine: iNCOVACC
- NISAR (NASA-ISRO Synthetic Aperture Radar) MISSION - February 10, 2023
- Lab Grown Diamonds - February 9, 2023
- Muons Particles - February 8, 2023
Intranasal COVID-19 vaccine: iNCOVACC
This article talks about How the Intranasal COVID-19 vaccine: iNCOVACC affects science and technology. In GS-3 and the following content has relevance for Daily Current Affairs for UPSC.
For prelims: Facts about Nasal vaccine
For mains: GS-3, Science and Biotechnology
Why in news:
- The Central Drugs Standard Control Organization of the Ministry of Health has approved the COVID-19 recombinant nasal vaccine for primary immunization of persons aged 18 and above in emergency situations.
- The vaccine is now only available to those who have not received vaccinations and goes by the name of iNCOVACC (BBV154).
About the Intranasal vaccine
- The most prevalent method of administering vaccines is intramuscular injections into the muscles or tissue adjacent to the muscles, which are generally used in a variety of methods (subcutaneous).
- A liquid solution can be administered orally rather than intravenously for some vaccines, for example, when administering to babies.

Intranasal COVID-19 vaccine: iNCOVACC
Mode of administration
- The vaccine is sprayed into the nostrils and inhaled nasally during intranasal administration.
- IgG, mucosal IgA, and T-cell responses are all neutralized when an intranasal vaccine elicits a wide immune response.
- The prevention of COVID-19 infection and transmission depends on immune responses at the injection site (in the nasal mucosa).
- The mucosa, the wet, spongy tissues that line the nose, mouth, lungs, and digestive tract, allows many viruses, including the coronavirus, to enter the body. This prompts these chemicals and cells to launch a particular immunological response.
- An intranasal vaccine, according to specialists, can combat the virus even before it tries to cross the body’s defenses.
- In most cases, intramuscular vaccines do not result in this mucosal response because they rely on immune cells that have been recruited from other parts of the body to swarm to the infection site.
Mechanism of action
- Through the B cells and T cells, which create immunoglobulins or antibodies like IgG to combat the disease-causing foreign invaders, vaccines typically elicit an immunological response in the body.
- By committing the foreign bodies to memory for future reaction, the T cells support the B cells.
- The B cells nearby the tissues of the mucosal lining will become active in response to nasal vaccinations.
- These B cells will generate IgA antibodies, which have been specifically designed to eliminate infections of the airways.
- To prepare for future responses, the local T cells will learn the pathogens.
Other Developments
- The vaccine will be produced across the nation, including in Gujarat, Karnataka, Maharashtra, and Telangana, and is stable between 2 and 8°C, making it simple to store and distribute.
- A novel intranasal SARS-CoV-2 vaccine, BBV154 or iNCOVACC, is an adenovirus vector with pre-fusion stabilized spike proteins.
- As a part of the third stimulus package, Atmanirbhar 3.0, the COVID Suraksha mission was established in order to support and expedite the development of the COVID-19 vaccine.
Mucosal vaccinations come in a wide variety, including:
- Single-dose nasal spray system: unit dose liquid
- Bidose nasal spray system: Bidose liquid
- The multi-dose liquid device system
- Nasal powder delivery technique that is an active and single dose
- Passive, nasal administration method for dose powder.
Benefits:
- The nasal route has significant potential for immunization due to the nasal mucosa’s well-organized immune system.
- It does not involve using any needles.
- Since it doesn’t require trained healthcare professionals, it is simple to administer.
- This can reduce hazards connected to using needles (injuries and infections).
- Both kids and adults will find it to be the perfect fit.
- The potential for scalable manufacturing will be able to satisfy the demand on a global scale.
- Special populations may benefit most from intranasal vaccinations:
- youngsters (simple to use, non-invasive) (easy to use, non-invasive)
- geriatric patients (easy to use, non-invasive)
- HIV-positive individuals (no fear of needle stick injuries)
- Those with many diseases (exhausted with injections)
Nasal vaccinations continue to have an advantage over intramuscular vaccines in terms of manufacture and transportation:
- The use of liquid and dry powder formulations is a definite benefit of intranasal administration.
- Because a cold chain may not be necessary, this benefits transportation and waste issues.
- It is possible to extend shelf life.
Concerns
- Until now, there hasn’t been much evidence to support the efficacy of this method of distribution, and attempts to do so haven’t worked well, with the exception of some flu vaccines.
- Although they are generally safe, it is difficult to attain their efficacy because it depends on several bioengineering techniques.
- Since there are still not enough human trial data available, widespread administration could result in erroneous results.
- The best vaccine protection comes from single-dosage devices, yet this type of filling technology is quite complex.
Globally Vaccine development programs:
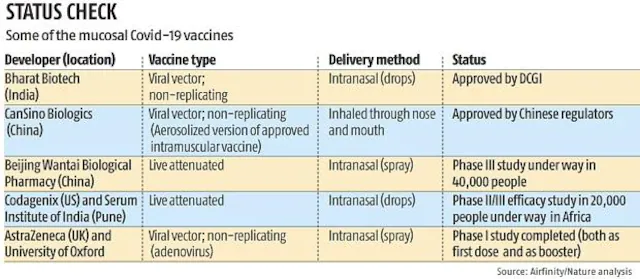
Globally Vaccine development programs
- Globally, there are about 100 COVID-19 mucosal vaccines under development. There are about 20 of them that have advanced to human clinical trials, and at least four of them have already finished or are now undertaking phase III investigations to gauge their safety and effectiveness in comparison to other vaccines.
- The delivery of at least five million doses of the vaccine approved for emergency use in Iran in October 2021. A viral vector-based nasal vaccination has also received approval from China.
- In September 2022, India also gave the internasal vaccination emergency approval.
- Vaccines like Covovax and Sputnik are currently accessible at private clinics, while the government’s Covid immunization campaign includes Covishield, Covaxin, and Corbevax.
Source:
Image Source:
Download PDF Now:
IAS Current Affairs
Current affairs are the main source of acquiring knowledge of the National and International World. So, Reading daily current affairs is very important for any competitive examination like UPSC, SSC, and other examinations.
Here Plutus IAS provides the latest and the best daily current affairs for the UPSC examination. So collect the daily, weekly, and monthly current affairs from the PlutusIAS current affairs site.




No Comments